【Column】Perspective of Biotech Consultant

Clinical Trials Demonstrating Superiority Can be Conducted Without Using Placebo
Author: Motoyoshi Okamura
●Domestic COVID-19 vaccine is aiming for early approval by conducting placebo-free trial
The Ministry of Health, Labor and Welfare has announced a policy to allow the use of clinical trial method to evaluate the efficacy of COVID-19 vaccine being developed in Japan without the use of placebo administration in order to speed up the practical use of the new vaccine. I suppose I am not the only one who thought “Is such a thing possible?” when hearing this news.
In order to prove the superiority of the new drugs, the clinical trials are divided into new drug administration group (SA: Study Arm) and placebo (dummy pill) administration group (CA: Control Arm), such as saline, and are in principle conducted in a so-called double-blind trial with random administration so that both patients and investigators do not know whether it is SA or CA.
The most time-consuming and expensive part of new drug development is clinical trials to evaluate its efficacy and safety. Especially in the case of cancer drugs, hundreds of test subjects are needed to prove the efficacy of a new drug, and half of the test subjects will be in the placebo administration group. It is natural for some patients to feel hesitate to participate in a clinical trial when they think that the investigational drug administered to them is a placebo with a 50% chance of not working at all, and the double-blind trial method have raised the ethical issue of “treating people as a guinea pig”. However, the double-blind trial method has undoubtedly been promoted as the standard clinical trial method in Japan and overseas because it is a statistically straightforward method for proving the superiority of a drug over a control group through trials.
The method of using a commercial vaccine as CA instead of a placebo, as in the present case, is a challenge but should be welcomed as a trial method that demonstrates the efficacy in that it follows the principles of statistics, however, a little more discussion is still needed before it can replace the current method.
●Biosimilar trials without using a placebo
However, this is not the first clinical trial to confirm efficacy without a placebo. Biosimilars trials are conducted in such way. Biosimilar trials make evaluations about the equivalence of a biosimilar to its predecessor. In other words, the biosimilar to be submitted is designated as
SA, and the predecessor is designated as CA, and the efficacy rates of the two administration groups are statistically compared for the primary endpoints (ACR20% improvement rate at 30 weeks in the example of rheumatoid arthritis trial as shown in the table). Clinical equivalence studies showed that the two groups were equivalent (no difference) in efficacy, which led to the approval of this biosimilar. Since no placebo was used in CA, subjects in both groups responded well to treatment of rheumatoid arthritis after being administered. Since there is no “failure” for the subjects regardless of whether they were administered SA or CA, they are strongly motivated to participate in a clinical trial that guarantees a cure for their disease, and as a result, more clinical evaluation data can be collected in a short period of time. If the preceding vaccine is used as CA in the clinical trial for the vaccine developed in Japan, it will be guaranteed that the vaccine will be equally effective in both groups, due to which a large number of subjects can be expected to be collected within a short period of time.
Examples of clinical trial results of biosimilar equivalence evaluation

(ACR20% improvement rate at 30 weeks)
●Non-inferiority trials and efficacy evaluation
A placebo is a dummy drug that contains no active ingredients and therefore has no medicinal effect at all. On the other hand, if an investigational drug containing active ingredients is administered, there will be a medicinal effect, and if its superiority over the placebo group is statistically evaluated, launching it as a new drug can be shown to be of greater significance. So what would be the method of evaluating efficacy in a clinical trial without a placebo? A non-inferiority trial is conducted to prove that the efficacy shown in the group being administered new vaccine is not inferior to that of the group being administered existing vaccine. The term “non-inferiority” gives a rather negative impression, and since we expect a new drug to be superior to the existing drugs, we want to show “superiority” rather than “inferiority,” so clinical trials to date have been conducted based on the theory that efficacy should be proven against a placebo, which has no medicinal effect apparently.
However, “non-inferiority” and “superiority” have different statistical interpretations. “Non-inferiority” is also different from “inferiority (significantly inferior efficacy) was not proven”. A margin of non-inferiority is established in a non-inferiority trial. As shown in the figure, SA and CA data of antibody titer after two vaccinations is obtained to test whether the estimated value of difference (SA-CA) (30 U/mL in the figure) is significant. In a superiority trial using placebo, if the 95% confidence interval exceeds the estimated value of the difference (30 in the figure), the conclusion can be drawn that “SA produced significantly higher antibody titers than CA”. On the other hand, as for non-inferiority trials, if the 95% confidence interval is not lower than the non-inferiority margin (-50 in the figure), it can be concluded that “efficacy is not inferior (equivalent) to CA”. Furthermore, in the case of equivalence evaluation of biosimilars, the confidence interval must fall within the equivalence margin (-50 to 50 in the figure), whereas in the non-inferiority trials, it only needs to prove that the confidence interval is greater than the non-inferiority margin. In other words, since non-inferiority trials do not take consideration of the upper limit, so even if a significantly higher antibody titer can be obtained from the new vaccine than the preceding one, the new vaccine will still meet the requirements for approval. The point is that if a clinical trial is conducted with the expectation of obtaining higher antibody titers than the preceding vaccine, and it shows significant superiority over CA as expected, it will bear the same meaning as the results of the current clinical trial using placebo.
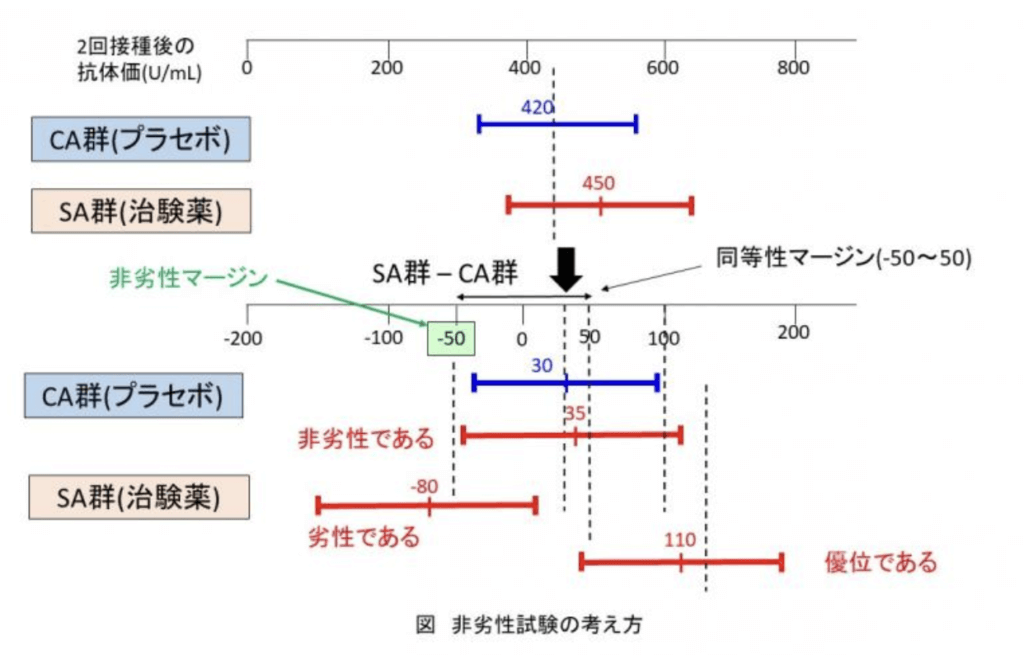
When considering why non-inferiority is acceptable, I believe that those in the position of developing new drugs must take into account the following that the author has written in Reference 2
“In the first place, since clinical trials aim to benefit patients, the primary endpoint should be a direct reflection of the clinical benefit to the patients in the final stage of a randomized phase III trial”
If it is a clinical trial conducted for the good of patients, a placebo trial should not be done. In the case of COVID-19 vaccine, even if the production of neutralizing antibodies, the primary endpoint, is inferior to that of the preceding vaccine, if it is possible to set, for example, the rate of reduction in aggravation risk due to vaccination as another primary endpoint, and if it is shown that SA is not inferior to CA in the rate of reduction in aggravation risk (including significantly superiority), the vaccine would meet the requirements for approval.
Although the Ministry of Health, Labor and Welfare is only considering the COVID-19 vaccine as a special exception and returning to conventional placebo-based trials once this pandemic is over, but on this occasion, I believe that non-inferiority trials without a placebo should be conducted in principle as an appropriate method of proving efficacy.
References
(1) Review Report May 8, 2014. Pharmaceuticals and Medical Devices Agency [infliximab subsequent 1]. (2) Written by Seiichi Satomi, supervised by Yoshimura Kenichi, The Correct Interpretation of Cancer Clinical Trials That No One Told You, Chugai Igaku-sha, 2011.
